 
I do find little about absorption spectrum of hydrogen molecule in the region 90 nm to 180 nm in some astrophysics research papers. In this model n = ∞ corresponds to the level where the energy holding the electron and the nucleus together is zero. The Balmer seriesthe spectral lines in the visible region of hydrogens emission spectrumcorresponds to electrons relaxing from n3-6 energy levels to the n2 energy level. Can somebody provide Absorption spectrum of Hydrogen molecule I have searched days for absorption spectrum of Hydrogen molecule in 200 nm - 300 nm UV region and All I got is absorption spectrum of atomic hydrogen. The calculated lowest energy transitions for these species were diagnostic of their protonation and oligomerization state. These states give rise to hydrogen-like series of absorption lines in the optical absorption spectrum of Cu2O at the photon energies described by the Rydberg. Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron.\) is the Rydberg constant in terms of energy, Z is the atom is the atomic number, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus. Recently we showed that visible-UV spectra in aqueous solution can be accurately calculated for arsenic (III) bisulfides, such as As(SH)3, As(SH)2S- and their oligomers. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. Stellar spectra are approximately a black body spectrum, but this mostly holds true for the most hot and luminous O and B type stars for cooler stars, the image already gets quite a bit more muddled, as they will show more and more absorption happening in the stellar photosphere, as well as other effects that ruin the nice, smooth black body sp. Wavelengths are in the ultraviolet region-13.6 eV 0.0 eV E PHYS 1493/1494/2699: Exp. n f 1 Unfortunately the Lyman series is not visible with the naked eye. The absorption spectra of the system under investi-gation show the presence of isobestic points which indicate the existence of 1: 1 hydrogen bonded complexes between OH group of phenols and the lone pair of electrons on nitrogen atom. The 4 lines of the visible hydrogen spectrum (the Balmer series) correspond to wavelengths of 410.2, 434.0, 486.1 and 656.3 nanometres (nm). 
The missing wavelengths tell us which wavelengths of the radiation are absorbed by the gas. absorption region in the range 250-290 nm. Bohr could now precisely describe the processes of absorption and emission in terms of electronic structure. 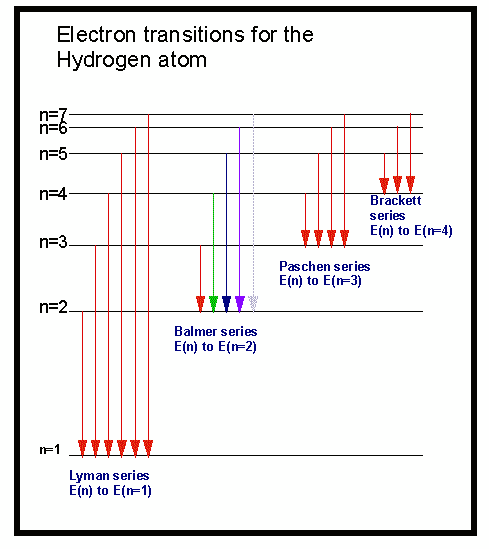
This spectrum appears as black lines that occur only at certain wavelengths on the background of the continuous spectrum of white light ( Figure 6.13 ). The Balmer seriesthe spectral lines in the visible region of hydrogen's emission spectrumcorresponds to electrons relaxing from n3-6 energy levels to the n2 energy level. The model could account for the emission spectrum of hydrogen and. In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. The emission spectrum of hydrogen Lyman series: It is made of all the de-excitations that end up on the n f 1 level Infinite number of them: n i 2, 3, 4. An absorption spectrum is observed when light passes through a gas. The model accounted for the absorption spectra of atoms but not for the emission spectra. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
